|
11/6/2023 0 Comments Heme bonded to co2 oxygen charge
The distal histidine, which is not bound to the heme, helps prevent oxidation of Fe2+ to Fe3+. It helps stabilize the position of the attached Heme. The affinity of CO to myoglobin-bound heme is only 250 times more than O2. Which of the following processes causes most of the carbon dioxide from the blood to move into the alveoli a.5 percent of oxygen in the blood is. The iron atom is bonded to a fifth nitrogen atom in the side chain of the deprotonated proximal histidine residue. On one side of the heme group is the proximal histidine, which binds the Fe2+ of the Heme to the nearby globin. charge or proton in distal Histidine makes a hydrogen bond with oxygen). Hemoglobin, or Hb, is a protein molecule found in red blood cells (erythrocytes) made of. The iron ion, which is the site of oxygen binding. Finally, this presentation aspires to lay a foundation for potential future clinical applications of these systems. The heme is located in a hydrophobic pocket of the protein. oxygen to cells and carbon dioxide to the lungs is (b) hemoglobin. A heme group consists of an iron (Fe) ion (charged atom) held in a heterocyclic ring, known as a porphyrin. Furthermore, the current understanding of the functional significance of HO shall be discussed from the perspective of each of the metabolic by-products, with a special emphasis on CO. 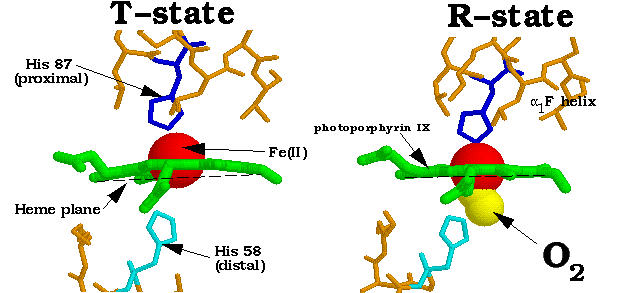
This review covers the molecular and biochemical characterization of HOs, with a discussion on the mechanisms of signal transduction and gene regulation that mediate the induction of HO-1 by environmental stress. The discovery that endogenously produced gaseous molecules such as NO and now CO can impart potent physiological and biological effector functions truly represented a paradigm shift and unraveled new avenues of intense investigations. The era of gaseous molecules in biomedical research and human diseases initiated with the discovery that the endothelial cell-derived relaxing factor was identical to the gaseous molecule nitric oxide (NO). In recent years, endogenously produced CO has been shown to possess intriguing signaling properties affecting numerous critical cellular functions including but not limited to inflammation, cellular proliferation, and apoptotic cell death. The heme oxygenases, which consist of constitutive and inducible isozymes (HO-1, HO-2), catalyze the rate-limiting step in the metabolic conversion of heme to the bile pigments (i.e., biliverdin and bilirubin) and thus constitute a major intracellular source of iron and carbon monoxide (CO). The Bader charge analysis further confirms that the adsorbed O 2 species possess a charge of 0.96e and the catalyst has a charge of +1.06e, which implies that the adsorbed oxygen species is in.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
